 
Elements from the same group of the periodic table show similar chemical characteristics. The rows of the table are called periods, and the columns are called groups. The table is divided into four roughly rectangular areas called blocks. It is a graphic formulation of the periodic law, which states that the properties of the chemical elements exhibit an approximate periodic dependence on their atomic numbers. 
It is widely used in chemistry, physics, and other sciences, and is generally seen as an icon of chemistry. Non-metals, on the other hand, rise from top to bottom of the periodic table.The periodic table, also known as the periodic table of the ( chemical) elements, is a rows and columns arrangement of the chemical elements. It gets smaller as we go from top to bottom. If the link between the atoms of elements is strong, it takes a lot of energy to break the bond. The melting point is the total amount of energy necessary to convert a solid into a liquid. As a result, we can deduce that as we move left to right in a period, the reactivity of elements steadily diminishes until group thirteen, when it begins to increase. Although the reactivity of nonmetals rises as one moves from left to right over time. 
As the metallic character decreases from left to right, so does the reactivity. As a result, the more metallic the element is, the more electropositive it is, and the more reactive it is. Metal reactivity is determined by its electropositive nature. The nonmetallic nature of a group of periodic table elements reduces as we proceed from top to bottom. The nonmetallic property of elements grows as we move left to right across a period in the periodic table. Non – Metallic Character of The Elements.The metallic property of elements grows as we progress up the periodic table from top to bottom. The metallic property of elements reduces as we move left to right across a period in the periodic table. The valency of elements from the same groups is the same. There is no change in valency among members of a group. Moving left to right across a period in the periodic table, valency increases first, then declines. Hydrogen, lithium, and sodium elements, for example, are all found in the first group and all have the same number of valence electrons, which is one.Īn atom’s valency is its ability to combine. It denotes that elements in the same group have the same number of valence electrons. Valence electrons remain consistent across a group. 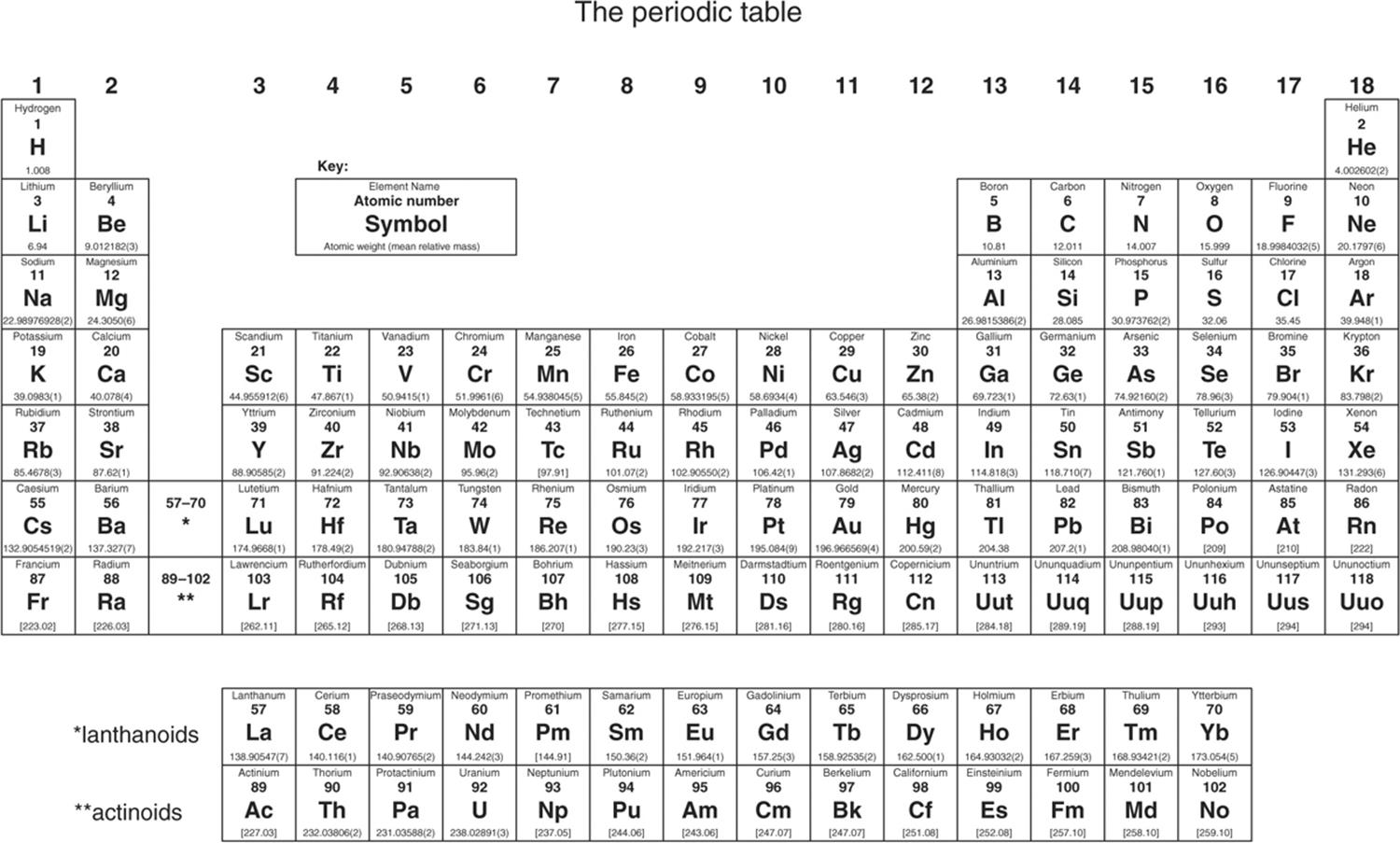
The amount of valence electrons grows as we move left to right across a period in the periodic table. The electrons in an atom’s outermost shell are referred to as the atom’s valence electrons. The explanation for this unusual behavior is that in the case of noble gases, atomic radius relates to van der Waal’s radius, whereas in the case of other elements, it refers to the covalent radius. When compared to the preceding halogen atom, the atomic radii of inter gases rapidly increase. ISRO CS Syllabus for Scientist/Engineer ExamĮxception: Noble gases exhibit extraordinary behavior.ISRO CS Original Papers and Official Keys.GATE CS Original Papers and Official Keys.DevOps Engineering - Planning to Production.Python Backend Development with Django(Live).Android App Development with Kotlin(Live).Full Stack Development with React & Node JS(Live).Java Programming - Beginner to Advanced.Data Structure & Algorithm-Self Paced(C++/JAVA).Data Structure & Algorithm Classes (Live). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
